
New Progress in Hydrogenation of Nitro Compounds over Diatomic Catalysts by ECUST Reported in Nature Communications
Recently, the Catalytic Reaction Engineering team from ECUST, led by Professor Xuezhi Duan and Researcher Xiaohu Ge, has achieved new progress in the development of catalysts for the hydrogenation of nitro compounds based on an adsorption-configuration matching strategy.
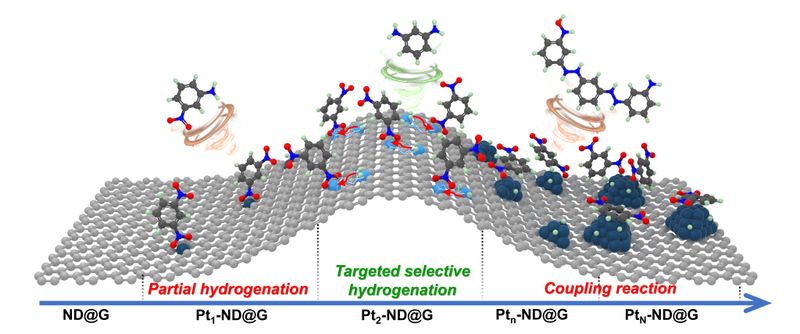
The team successfully constructed a diatomic Pt₂2-ND@G catalysts with high-density and non-bonding structural characteristics, which realized the spatial decoupling of nitro compound adsorption and hydrogen activation sites, thereby overcoming the long-standing trade-off between catalytic activity and selectivity in hydrogenation reactions. The findings were published in Nature Communications under the title “Synergistic non-bonding diatomic Pt sites for efficient hydrogenation of nitro compounds.”
Hydrogenation of nitro compounds is a core process in the manufacture of high-value chemicals. Its key challenge lies in achieving the selective activation of nitro groups within multifunctional molecules for the directed synthesis of important intermediates such as phenylenediamines.
Although supported platinum-group metal catalysts exhibit excellent hydrogenation activity due to their optimized d-band structure, the stepwise reduction of nitro groups leads to electronic shielding and steric hindrance, which inhibit subsequent intermediate transformation.
As a result, it is difficult to simultaneously achieve high activity and high selectivity toward target products. Previous studies have shown that tuning active sites to an atomically dispersed state can significantly improve metal utilization efficiency. However, the limited capability of isolated sites for hydrogen dissociation often becomes a key bottleneck restricting catalytic performance (AIChE J. 2024, 70, e18278; ACS Catal. 2025, 15(14), 12365).
To address this challenge, the research team employed a synergistic diatomic-site catalysis strategy. By constructing high-density, electron-deficient Pt₂ sites with non-bonding characteristics on a nanodiamond-graphene (ND@G) support, this design achieved effective decoupling of nitro compound adsorption and hydrogen activation, significantly enhancing the selective hydrogenation performance.
Specifically, the team utilized a two-step atomic layer deposition method to anchor high-density non-bonding diatomic Pt sites in a controlled manner onto a defect-rich, thermally induced surface-modified nanodiamond–graphene support. Aberration-corrected high-resolution scanning transmission electron microscopy and X-ray absorption spectroscopy confirmed that the Pt species predominantly exist as non-bonding diatomic pairs with an interatomic distance of approximately 0.41 nm, exhibiting pronounced electronic coupling characteristics.
In-situ infrared spectroscopy and theoretical calculations reveal that this unique spatial proximity effect of the diatomic sites not only enables effective site decoupling between nitro adsorption and hydrogen activation, but also significantly weakens the adsorption strength of target product molecules. Catalytic performance evaluation demonstrated that the diatomic catalyst achieves complete conversion of nitro compounds under mild conditions with over 99.0% selectivity toward the desired products, outperforming previously reported platinum-group metal catalysts.
The co-first authors are Minhao Chen and Yundao Jing (PhD candidates). The corresponding authors are Professor Xuezhi Duan and Researcher Xiaohu Ge from ECUST. The research was conducted under the guidance of Academicians Weikang Yuan and De Chen, as well as Professor Xinggui Zhou.
This work was supported by the National Key R&D Program of China, the National Natural Science Foundation of China, as well as projects funded by the Shanghai Municipal Commission of Education and the Shanghai Municipal Commission of Science and Technology.