
New Progress in Electrocatalytic Interfacial Microenvironment Regulation from ECUST Published in Accounts of Chemical Research
Recently, a research team led by Professor Chunzhong Li from the School of Chemical Engineering at ECUST published a review article titled “Mastering the Electrified Interface Microenvironment for Selective Electrocatalysis” in Accounts of Chemical Research. The article summarized the advances and perspectives on electrocatalytic interfacial microenvironment regulation.
Electrocatalysis is often limited by side reactions and mass transport constraints arising from poorly controlled electrode–electrolyte interfaces. While conventional approaches mainly focus on optimizing intrinsic catalyst activity through composition and morphology, the team emphasized that the interfacial microenvironment critically governs reaction selectivity and kinetics.
Their research demonstrated that even seemingly identical catalysts could exhibit significantly different electrocatalytic performance under slightly altered electrolyte conditions. These differences could be attributed to changes in proton reactivity, ion distribution, and solvent structure at the interface. Based on these insights, the team argued that rational electrocatalyst design must be integrated with deliberate engineering of the interfacial microenvironment.
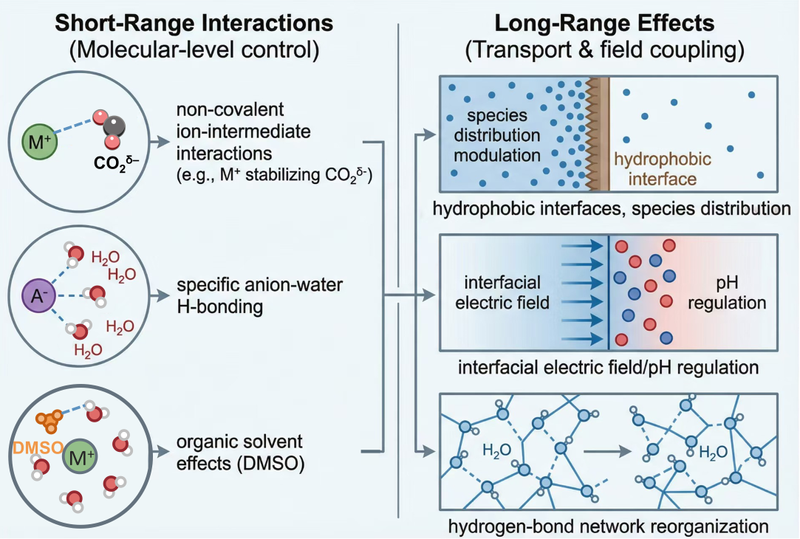
The article focused on three representative reactions: CO2 reduction (CO2RR), oxygen reduction (ORR), and organic hydrogenation. The researchers distinguished between short-range interactions, such as noncovalent ion–intermediate coordination and hydrogen bonding, and long-range effects, including interfacial pH gradients and electric field distributions. Together, these interactions and effects regulate reaction pathways and the concentration profiles of reactants and protons.
To establish structure-activity-performance relationships, the team combined operando characterization with multiscale simulation, including in situ vibrational spectroscopy, electrochemical impedance spectroscopy (EIS), density functional theory (DFT), and ab initio molecular dynamics (AIMD).
The study showed that electrolyte components, including alkali and organic cations, functional anions, organic cosolvents, surfactants, and polymers, can be used to reshape the interfacial microenvironment and steer proton-coupled electron transfer (PCET) pathways. This enabled selective production of valuable products while suppressing competing reactions.
This work provided a mechanism-driven framework for designing next-generation electrochemical systems and advancing carbon-neutral energy sustainable technologies.
The first author of the paper is Dr. Wangxin Ge from the School of Chemical Engineering at ECUST, and the corresponding authors are Professor Chunzhong Li and Professor Hongliang Jiang. This work has been supported by the National Natural Science Foundation of China, the China Postdoctoral Science Foundation, and the Shanghai Municipal Science and Technology Major Project.